This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison
Small Molecule Interactions
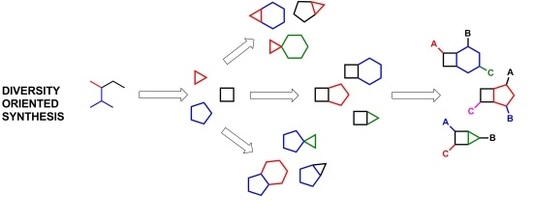
Small molecules are often used in chemical assays to determine their affinity to a protein of interest. They can be used to characterize the mechanisms of interaction in proteins that are not well-studied, as well as find potential drugs that can be used as protein inhibitors. The knowledge about the protein determines the type of small molecule library that is used. When the protein is first being characterized and all of its functions are still being determined, a diversity-oriented library is often used to test a broad variety of small molecules and determine what classes show the highest affinity to the protein on interest. These molecules are highly variable and designed to present as many types of molecules to the protein as possible [1].
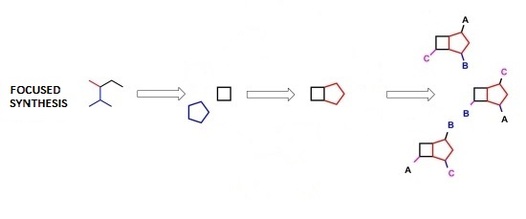
If more finesse is desired, a target-oriented (biologically-oriented) library can be used in which many molecules with very similar structures are introduced to the protein to see what variation of the base molecule has the highest affinity. This includes molecules with the same skeleton but often differing in their individual functional groups. Often these types of libraries lead to the discovery of many drugs that effectively act on a protein but vary in their side effects or strength [1].
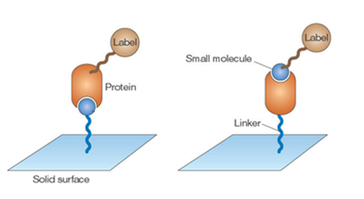
Analysis from this point can continue by immobilizing either the small molecule library or the protein of interest on a solid surface with a linker. The other is labeled with a fluorescent marker and put into solution. The solution is washed over the solid surface so that any binding partners will interact with each other and the rest of the solution can be washed away. All molecule-protein binding sites will fluoresce to indicate the space on the solid surface where the interaction is taking place. Those small molecules can be collected, identified using mass spectrometry, and further tested to determine their overall effects on the protein of interest [1].
Analysis
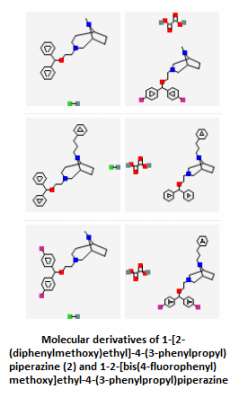
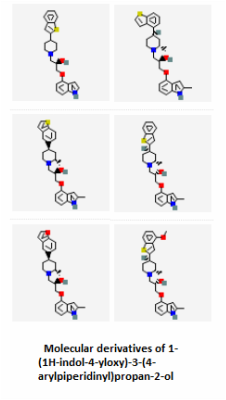
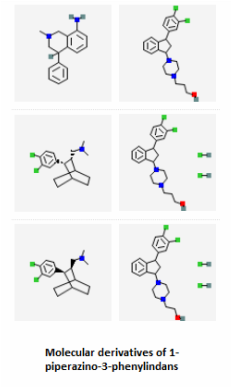
The PubChem bioassays database was used to find small molecules that have been tested against the SLC6A4 protein in various experiments. A total of 8,654 results were returned, but 3,196 of those inactive or unspecified. Most of these molecules were being tested as potential serotonin reuptake inhibitors for treating anxiety and mood disorders such as OCD or depression. Several examples are shown below.
Discussion
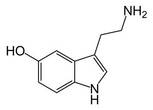
Serotonin reuptake inhibitors (SRIs) have commonly been used as treatments for OCD [2], so the fact that there are thousands of active molecules binding SLC6A4 that are related to serotonin reuptake inhibition lends great support to the treatment. Those compounds shown above are just a fraction of the small molecules that can possibly used to interact with SLC6A4. Notice how similar several of the molecules are to the actual structure of serotonin itself. Aromatic rings are common features in each of the active small molecules, and some even go so far as to include the fused ring structure that is the base of the serotonin skeleton. Because so much has already been studied in terms of SRIs and their functions, it appeared on PubChem that target-oriented libraries rather than diversity-oriented libraries have been used more commonly to find the best candidates for new SRIs. These new drugs can then be used to give some relief to people with mood and anxiety disorders like depression or OCD.
References
PubChem: http://pubchem.ncbi.nlm.nih.gov/search/
[1] O'connor, C., Laraia, L., & Spring, D. (2011). Chemical Genetics. Chem. Soc. Rev., 40, 4332-4345. Retrieved March 23, 2015, from http://pubs.rsc.org/en/Content/ArticleLanding/2011/CS/c1cs15053g#!divAbstract
[2] How is OCD Treated? (2014, April 28). Retrieved February 19, 2015, from http://iocdf.org/about-ocd/treatment/
Pictures from top to bottom:
http://www.mdpi.com/1420-3049/19/10/16506
http://www.mdpi.com/1420-3049/19/10/16506 (with modifications made in Paint)
http://www.nature.com/nature/journal/v432/n7019/images/nature03196-f2.2.jpg
http://commons.wikimedia.org/wiki/File:Serotonin-skeletal.png
Small Molecule Images:
Small Molecules 1-6: Zhang, Y., Rothman, R., Dersch, C., Costa, B., Jacobson, A., & Rice, K. (2000). Synthesis and Transporter Binding Properties of Bridged Piperazine Analogues of 1-{2-[Bis(4-fluorophenyl)methoxy]ethyl}-4-(3-phenylpropyl)piperazine (GBR 12909). J. Med. Chem., 43(25), 4840-4849. Retrieved March 23, 2015, from http://pubs.acs.org.ezproxy.library.wisc.edu/doi/abs/10.1021/jm000300r
Small Molecules 7-12: Rocco, V., Spinazze, P., Kohn, T., Honigschmidt, N., Nelson, D., Wainscott, D., ... Takeuchi, K. (2004). Advances toward new antidepressants beyond SSRIs: 1-aryloxy-3-piperidinylpropan-2-ols with dual 5-HT1A receptor antagonism/SSRI activities. Part 4. Bioorganic & Medicinal Chemistry Letters, 14 17 May 2004, Pages 2653–2656(10), 2653-2656. Retrieved March 23, 2015, from http://www.sciencedirect.com.ezproxy.library.wisc.edu/science/article/pii/S0960894X0400304X
Small Molecules 13-18: Huffman, W., Hall, R., Grant, J., Wilson, J., Hieble, J., & Hahn, R. (1983). 4-(Aminoalkyl)-7-hydroxy-2(3H)-indolones, a novel class of potent presynaptic dopamine receptor agonists. Journal of Medicinal Chemistry, 26(7), 933-935. Retrieved March 24, 2015, from http://pubs.acs.org.ezproxy.library.wisc.edu/toc/jmcmar/26/7
[1] O'connor, C., Laraia, L., & Spring, D. (2011). Chemical Genetics. Chem. Soc. Rev., 40, 4332-4345. Retrieved March 23, 2015, from http://pubs.rsc.org/en/Content/ArticleLanding/2011/CS/c1cs15053g#!divAbstract
[2] How is OCD Treated? (2014, April 28). Retrieved February 19, 2015, from http://iocdf.org/about-ocd/treatment/
Pictures from top to bottom:
http://www.mdpi.com/1420-3049/19/10/16506
http://www.mdpi.com/1420-3049/19/10/16506 (with modifications made in Paint)
http://www.nature.com/nature/journal/v432/n7019/images/nature03196-f2.2.jpg
http://commons.wikimedia.org/wiki/File:Serotonin-skeletal.png
Small Molecule Images:
Small Molecules 1-6: Zhang, Y., Rothman, R., Dersch, C., Costa, B., Jacobson, A., & Rice, K. (2000). Synthesis and Transporter Binding Properties of Bridged Piperazine Analogues of 1-{2-[Bis(4-fluorophenyl)methoxy]ethyl}-4-(3-phenylpropyl)piperazine (GBR 12909). J. Med. Chem., 43(25), 4840-4849. Retrieved March 23, 2015, from http://pubs.acs.org.ezproxy.library.wisc.edu/doi/abs/10.1021/jm000300r
Small Molecules 7-12: Rocco, V., Spinazze, P., Kohn, T., Honigschmidt, N., Nelson, D., Wainscott, D., ... Takeuchi, K. (2004). Advances toward new antidepressants beyond SSRIs: 1-aryloxy-3-piperidinylpropan-2-ols with dual 5-HT1A receptor antagonism/SSRI activities. Part 4. Bioorganic & Medicinal Chemistry Letters, 14 17 May 2004, Pages 2653–2656(10), 2653-2656. Retrieved March 23, 2015, from http://www.sciencedirect.com.ezproxy.library.wisc.edu/science/article/pii/S0960894X0400304X
Small Molecules 13-18: Huffman, W., Hall, R., Grant, J., Wilson, J., Hieble, J., & Hahn, R. (1983). 4-(Aminoalkyl)-7-hydroxy-2(3H)-indolones, a novel class of potent presynaptic dopamine receptor agonists. Journal of Medicinal Chemistry, 26(7), 933-935. Retrieved March 24, 2015, from http://pubs.acs.org.ezproxy.library.wisc.edu/toc/jmcmar/26/7